|
Biomimetic total synthesis of berkeleyamide D is described. In 2004, Snider and Neubert reported a novel biomimetic route to 3-acyl-5-hydroxy-3-pyrrolin-2-ones.1 Inspired by this biomimetic sequence, our group envisioned a biosynthetically inspired total synthesis of berkeleyamide D. We sought to construct the oxa-azaspirocyclic core of berkeleyamide D from a linear precursor via a series of oxidation processes. Our synthetic strategy forms the basis for the synthesis of various natural products which contain the oxa-azaspirononenedione core. |
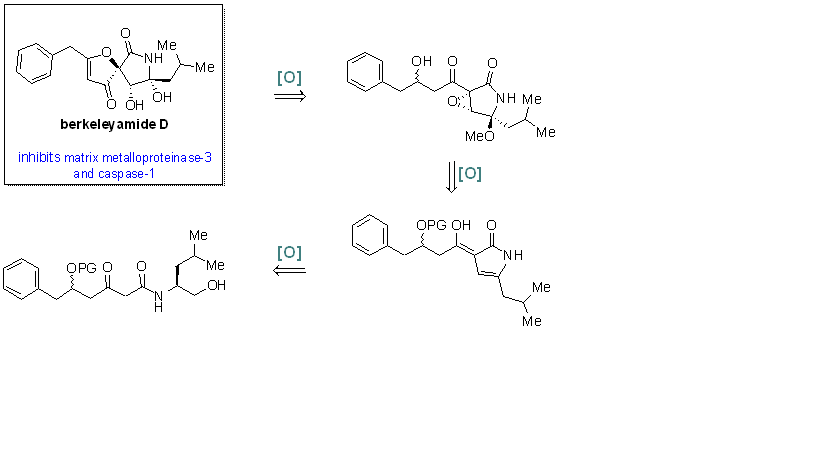
|
 119th General Meeting of the KCS
119th General Meeting of the KCS
 119th General Meeting of the KCS
119th General Meeting of the KCS