|
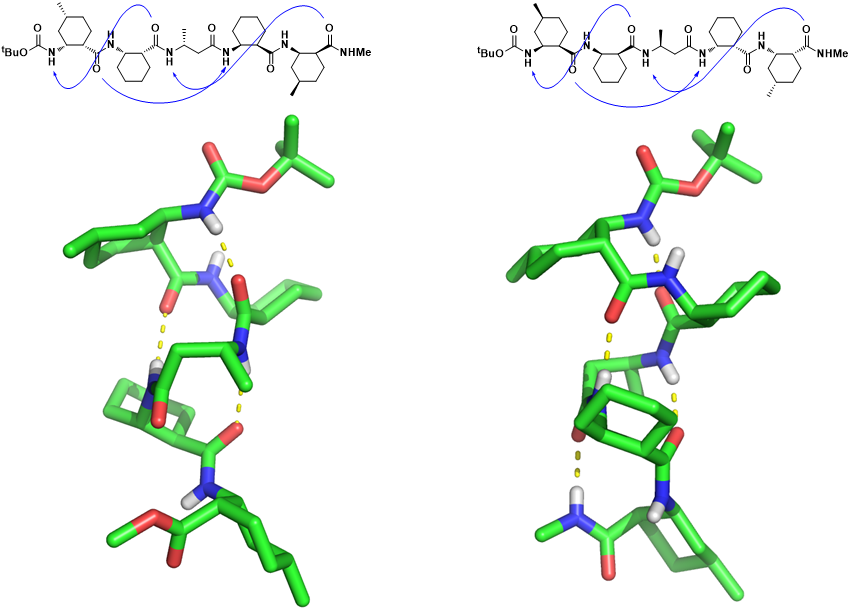
Within the past two decades, the folding properties of several types of foldamers with unnatural backbones have been explored. Among them, β-peptides adopting well-defined secondary structures are particularly interesting as scaffolds. Herein we report that 12/10-helical propensity is well-maintained after partial replacement of cyclic β-amino acid residue with acyclic β3-amino acid residue known to be quite malleable. In those 12/10-helices, unlike in earlier studies, acyclic L-β3-residue has an unusual gauche(-) conformation in spite of steric strain, which is derived from high-resolution crystal structures. Our results show that acyclic β3-amino acid residue can be successfully incorporated into the 12/10-helical β-peptide if most of the residues are appropriately pre-organized for folding, even though those β3-residues have energetically unfavorable conformations. We hope that these results can enrich the domain of β-peptide foldamer chemistry. |
|
 119th General Meeting of the KCS
119th General Meeting of the KCS
 119th General Meeting of the KCS
119th General Meeting of the KCS