|
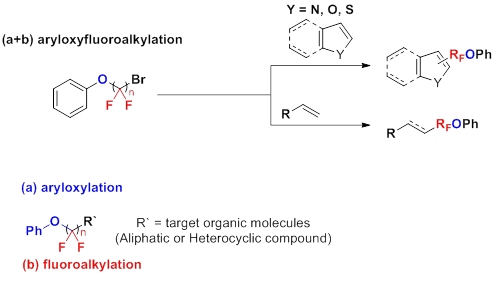
Installation of fluoroalkyl groups has been of great interest in many fields because of their ability to change the physical, chemical, and biological properties such as lipophilicity, binding selectivity, metabolic stability and bioavailability. In particular, the introduction of fluoroalkyl groups containing oxygen has been of growing interest because the presence of oxygen can further change the properties of molecules.
We have developed a visible light-induced oxyfluoroalkylations under mild reaction conditions. By this approach, two functional groups consisting of oxygen and fluoaoalkyl moieties could be simultaneously installed to various organic molecules, showing the potential use of the method for late-stage modifications in the development of functional molecules. In addition, this method can be easily utilized for fine-tuning of properties in drug development by controlling the number of fluorine atoms in reagents
|
|
 119th General Meeting of the KCS
119th General Meeting of the KCS
 119th General Meeting of the KCS
119th General Meeting of the KCS