|
Polycyclic aromatic and heteroaromatic compound have received tremendous attention due to their various biological, electronic properties and have been utilized as the important structural motifs in many applications. In particular, because of their strong fluorescence emission property, they have been applied to various electronic devices such as OLEDs, OFETs.
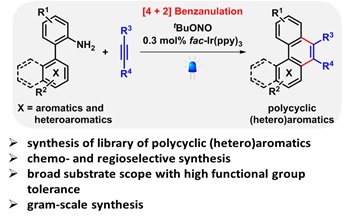
We have synthesized a library of polycyclic aromatic and heteroaromatic compound through visible-light induced [4+2] benzannulations between 2-(hetero)aryl-substituted anilines and (hetero)aryl alkynes in the presence of fac-Ir(ppy)3 as the photocatalyst and tBuONO as the diazotization reagent. The in situ generation of diazonium salt as intermediate allowed extended substrate scope that have not been easy to access. In this talk, the detailed study on chemo- and regio-selective [4+2]-benzannulation to synthesize highly substituted tricyclic- and tetracyclic compounds including phenanthrene derivatives will be presented.
|
|
 119th General Meeting of the KCS
119th General Meeting of the KCS
 119th General Meeting of the KCS
119th General Meeting of the KCS