|

|
| Type |
Poster Presentation |
| Area |
Organic Chemistry |
| Room No. |
Exhibition Hall 2+3 |
| Time |
10월 20일 (금요일) 13:00~14:30 |
| Code |
ORGN.P-253 |
| Subject |
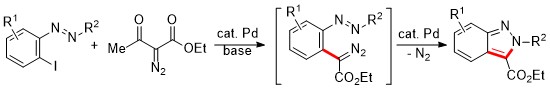
Synthesis of 2H-Indazoles via Palladium-Catalyzed Deacylative Cross-Coupling and Denitrogenative Cyclization from 2-Iodoazoarenes and 2-Iodoaryltriazenes |
| Authors |
Gi Uk Han, Kyusik Um, Dongjin Kang1, Phil Ho Lee*
Department of Chemistry, Kangwon National University, Korea
1Department of Pharmaceutics, Inje University, Korea
|
| Abstract |
|
A synthetic method to prepare a wide range of 2H-indazoles was developed via a tandem Pd-catalyzed deacylative cross-coupling reaction of 2-iodoazoarenes and 2-iodoaryltriazenes with acyldiazoacetates and denitrogenative cyclization reaction of in situ generated diazoacetates having azoaryl and triazenyl aryl moieties in one-pot. Additionally, azoaryl-substituted diazoacetates underwent Pd-catalyzed denitrogenative cyclization to produce 2H-indazoles. The present reaction is a good example in which a Pd(0)-catalyst is involved in two catalytic cycles in one-pot.This work was supported by the Human Resource Training Program for Regional Innovation and Creativity through the Ministry of Education and National Research Foundation of Korea (NRF-2015H1C1A1035955) |
|
|
|
| E-mail |
gksrldnr91@naver.com |
|
 120th General Meeting of the KCS
120th General Meeting of the KCS
 120th General Meeting of the KCS
120th General Meeting of the KCS