|

|
| Type |
Poster Presentation |
| Area |
Organic Chemistry |
| Room No. |
Exhibition Hall 2+3 |
| Time |
10월 20일 (금요일) 13:00~14:30 |
| Code |
ORGN.P-267 |
| Subject |
Preparation of 1-(t-butyl)dimethylsilyl-2,2-difluorostyrenes |
| Authors |
SEOHEE LEE, In Howa Jeong1,*
Chemistry, Yonsei University, Korea
1Chemistry and Department of Medical Chemistry, Yonsei University, Korea
|
| Abstract |
|
1-(t-Butyl)dimethylsilyl-2,2-difluoroethenyl tosylate (2) was prepared in 83% yield from the reaction of 2,2,2-trifluoroethyl tosylate (1) with 2.2 equiv of LDA in THF at -78 ℃, followed by treatment with (t-butyl)dimethylsilyl chloride (1.0 equiv). Cross-coupling reaction of 2 with arylboronic acid (2.0 equiv) in the presence of Pd2(dba)3 (10 mol%), SPhos (20 mol%) and Cs2CO3 (1.7 equiv) in a cosolvent of dioxane and H2O (10:1) at 100 ℃ for 6-10 hours afforded the coupled product 3 in moderate to good yields. The scopes and limitations of this reaction will be presented. |
|
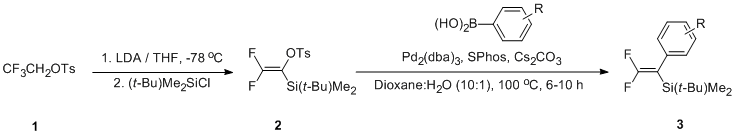
|
|
| E-mail |
godgod0518@naver.com |
|
 120th General Meeting of the KCS
120th General Meeting of the KCS
 120th General Meeting of the KCS
120th General Meeting of the KCS