|
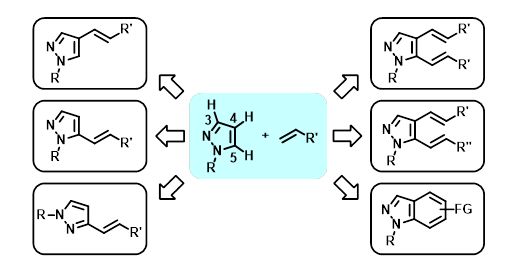
We have developed Pd-catalyzed regioselective C4-, C5-, and di-alkenylation of pyrazoles. Kinetic isotope effects and H/D exchange experiments were performed in order to obtain mechanistic insights into C4- and C5-alkenylation. These Pd-catalyzed protocols gave a wide range of alkenylated pyrazoles from readily available, inexpensive pyrazoles and alkenes. In addition, thermal 6-electrocyclization and oxidation via dialkenylated pyrazoles provided functionalized indazoles, which have important roles in medicinal chemistry and material science. Furthermore, C5-alkenyl pyrazoles were useful for the preparation of 3-alkenyl pyrazoles that are not easily accessible by C−H alkenylation. This comprehensive strategy can provide a variety of pyrazole and indazole derivatives for applications in the development of biologically active compounds and
]functional materials.
|
|
 120th General Meeting of the KCS
120th General Meeting of the KCS
 120th General Meeting of the KCS
120th General Meeting of the KCS