|
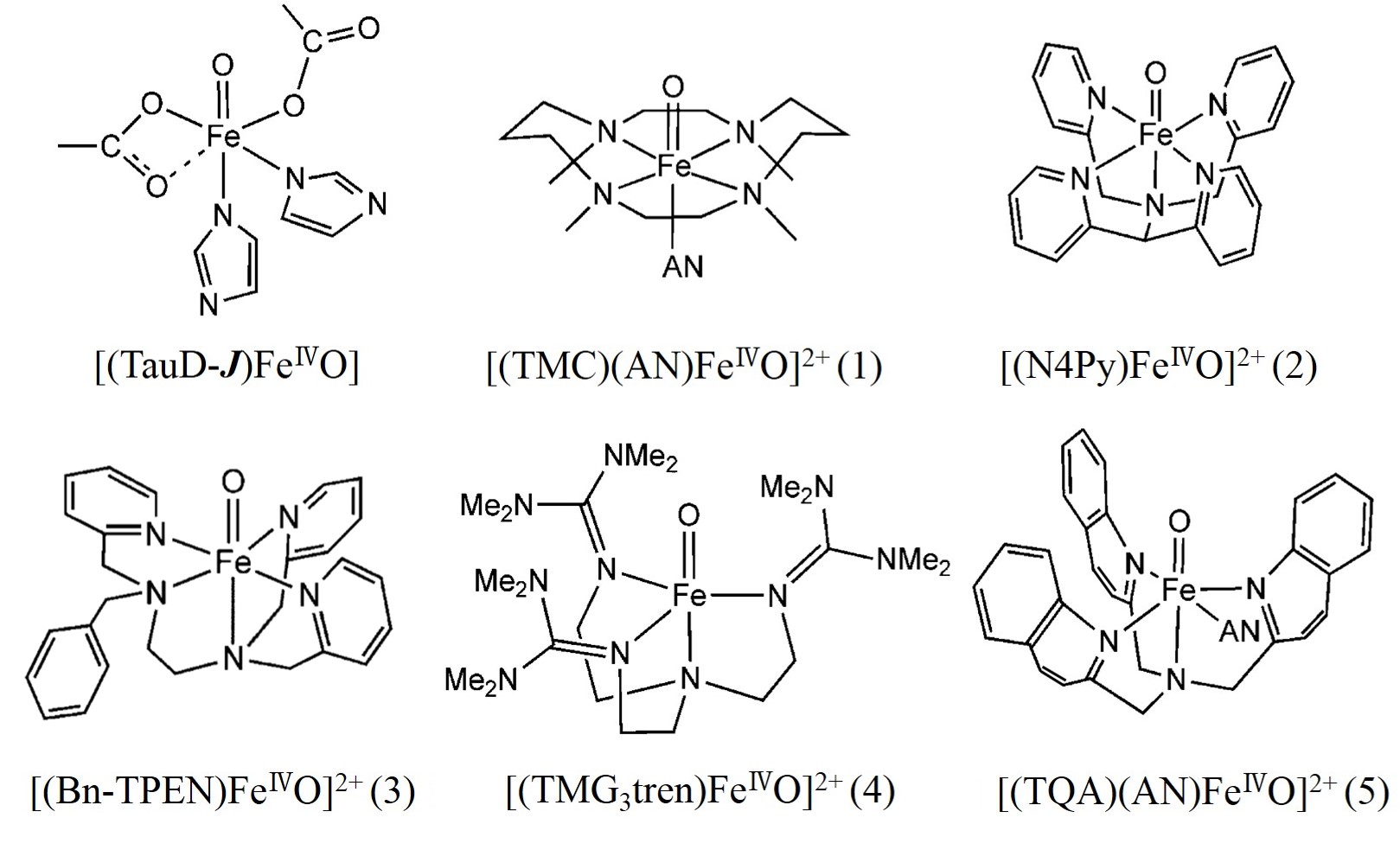
High valent FeIVO species has been proposed to play a key role in the catalytic cycle of non-heme mono-iron enzymes. Extensive studies have been performed to reveal the catalytic mechanism of the enzyme and to reproduce its catalytic reactivity using biomimetic FeIVO complexes with synthetic ligands. In this study, the mechanism of C-H activation has been investigated at an atomic detail for four synthetic complexes with an Oh symmetry and two with a Tbp symmetry including a TauD-J model (see below), based on the DFT potential energy surfaces of both S =1 and S =2 spin states. This study demonstrated that all the high spin FeIVO complexes, irrespective of their symmetry (Tbp or Oh), utilize a species like an FeIII-oxyl radical for C-H activation, which is formed en route to the TS before the actual HAT occurs. However, no such species is formed in the intermediate spin state (S = 1). The reason why the reactivity of high spin complexes depends on ligand symmetry even with nearly identical mechanism will be discussed.
|
|
 120th General Meeting of the KCS
120th General Meeting of the KCS
 120th General Meeting of the KCS
120th General Meeting of the KCS