|

|
| Type |
Poster Presentation |
| Area |
Medicinal Chemistry |
| Room No. |
Exhibition Hall 2+3 |
| Time |
10월 19일 (목요일) 11:00~12:30 |
| Code |
MEDI.P-314 |
| Subject |
Anticancer and Analgesic Activities of Fluoro-substituted 3,4-Dihydroquinazoline Derivatives |
| Authors |
Da Woon Jung, Hong bin Yoon1, Jae Yeol Lee1,*
Chemistry, Kyung Hee University, Korea
1Department of Chemistry, Kyung Hee University, Korea
|
| Abstract |
|
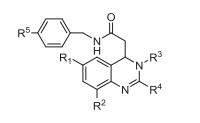
As a bioisosteric strategy to overcome the poor metabolic stability of lead compound KYS05090S, a series of new fluoro-substituted 3,4-dihydroquinazoline derivatives was prepared and evaluated for T-type calcium channel (Cav3.2) block, cytotoxic effects and liver microsomal stability. Among them, compound 8h (KCP10068F) containing 4-fluorobenzyl amide and 4-cyclohexylphenyl ring potently blocked Cav3.2 currents (>90% inhibition) at 10 μM concentration and exhibited cytotoxic effect (IC50 = 5.9 μM) in A549 non-small cell lung cancer cells that was comparable to KYS05090S. Furthermore, 8h showed approximately a 2-fold increase in liver metabolic stability in rat and human species compared to KYS05090S. Based on these overall results, 8h (KCP10068F) may therefore represent a good backup compound for KYS05090S for further biological investigations as novel cytotoxic agent. In addition, compound 8g (KCP10067F) was found to partially protect from inflammatory pain via a blockade of Cav3.2 channels. |
|
|
| E-mail |
jdw0622@naver.com |
|
 120th General Meeting of the KCS
120th General Meeting of the KCS
 120th General Meeting of the KCS
120th General Meeting of the KCS