|
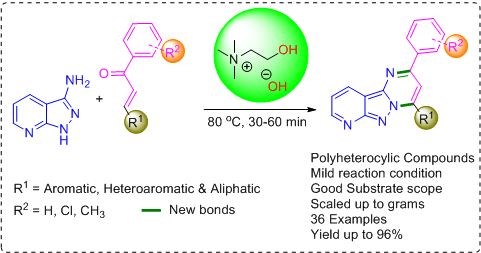
Choline hydroxide has been found to be a green and efficient basic ionic liquid catalyst for the synthesis of highly functionalized pyrido[2',3':3,4]pyrazolo[1,5-a]pyrimidine derivatives through the reaction of a series of α,β-unsaturated ketones with 1H‐Pyrazolo[3,4‐b]pyridin‐3‐amine under neat conditions. A series of α,β-unsaturated ketones with different substituted functional groups efficiently converted to their corresponding products in good to excellent isolated yields, and the reactions can be easily scaled up to multigrams. The present environmentally benign and reusable ChOH catalyst offers several advantages such as shorter reaction times, a wide range of functional group tolerance, and high yield of products via a simple experimental and work-up procedure. |
|
 121st General Meeting of the KCS
121st General Meeting of the KCS
 121st General Meeting of the KCS
121st General Meeting of the KCS