|
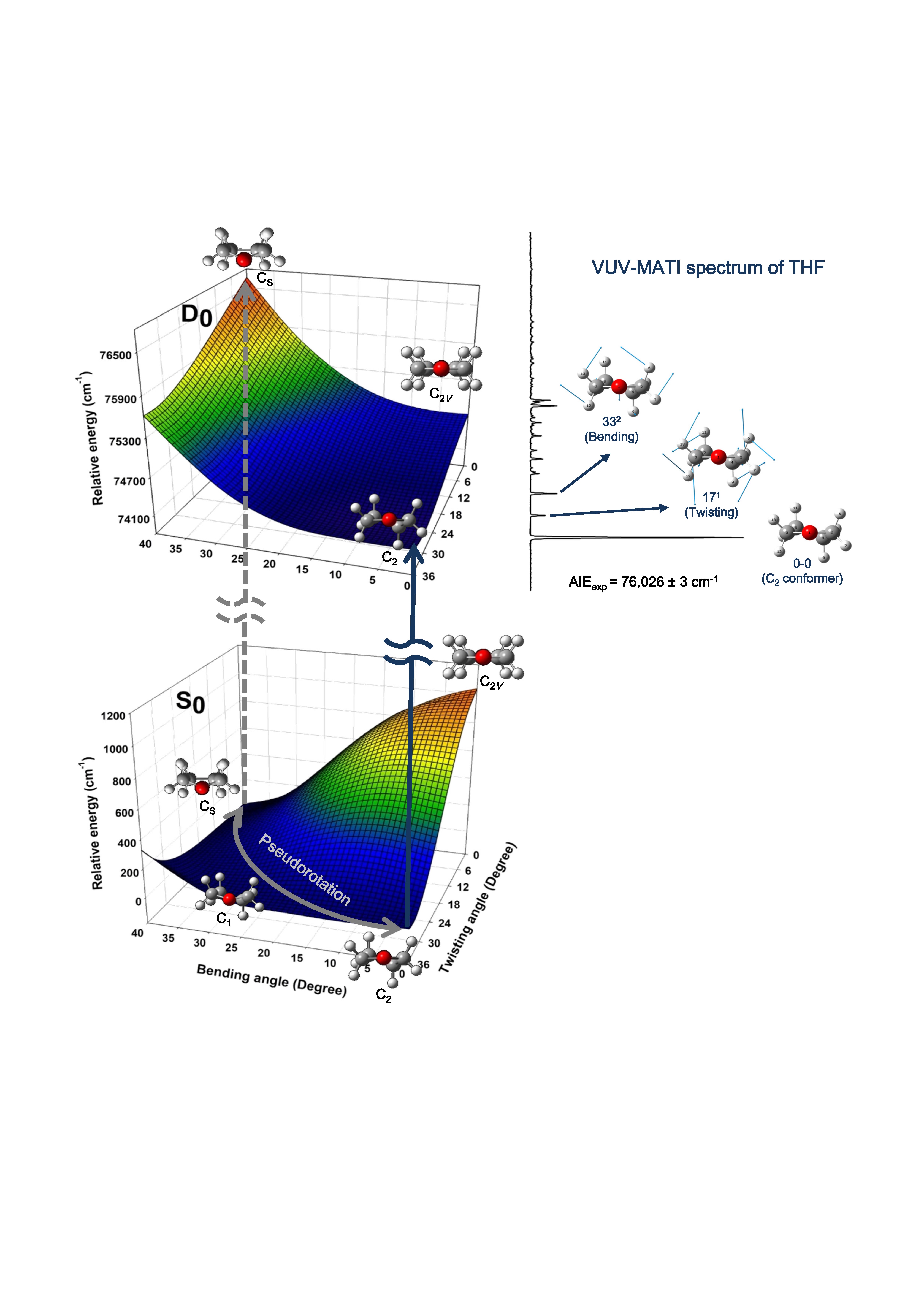
We investigated the essential conformations of heterocyclic molecular cation employing new spectroscopic technique with spectrometers based on mass spectrometry and laser spectroscopy in combination with quantum chemical calculations, since heterocyclic ring compounds constituting the backbone of large biomolecules generally have the biological activities depending on conformational structure. The high-resolution vacuum ultraviolet mass-analyzed threshold ionization (VUV-MATI) spectroscopy was firstly used to determine the accurate ionization energies of thietane, oxatane, tetrahydrothiophene, and tetrahydrofuran and measure the vibrational spectra of their cations. We found that the conformations of saturated four-membered rings generally reflect the balance between ring strain and torsional forces arising from non-tetrahedral methylene groups, which affects ring-puckering vibrations. For the five-membered rings, the conformational changes of cyclic compounds experiencing large torsional forces can occur along with ring-puckering vibrational motion with the puckering phase rotating around the ring, which is denoted as pseudorotation. |
|
 121st General Meeting of the KCS
121st General Meeting of the KCS
 121st General Meeting of the KCS
121st General Meeting of the KCS