|
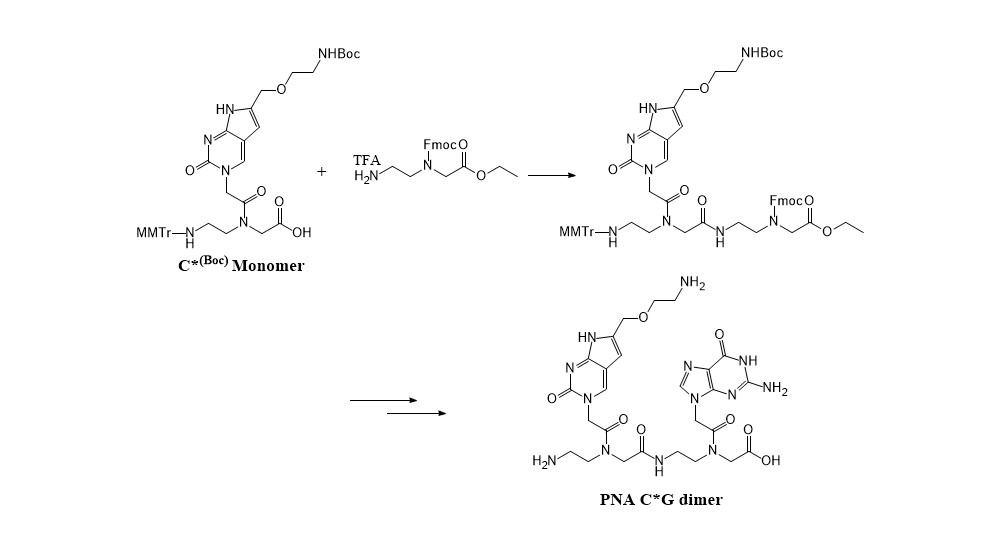
Recently Israel researchers have found that a PNA dimer forms a specific quaternary structure by self-replication of bases at a specific pH and exhibits a unique fluorescence property, which can be used as an OLED device. This result is significant in that PNA has a new application for an electronic material in addition to the biological field. To further investigate the applicability of PNA as an electronic material, we tried to make a PNA dimer with a modified cytosine base (C*). The C* was designed to have strong complementary bonds by G-clamp formation. Therefore, we aimed to investigate the effect of modified cytosine on the self-replication pattern and fluorescence properties. For this purpose, we needed to make C*G and GC* PNA dimers. However, the synthesis of di-PNA containing modified cytosine required a quite different synthetic strategy than the conventional PNA synthesis method in order to minimize the acyl migration and improve the yield of coupling process between PNA monomers. We found MMTr N-terminal protection was the best to minimize the acyl migration side reaction. Adding the 2nd base later after attaching the PNA unit first led to a significant improvement in the yield, and finally the desired PNA dimer C*G and GC* could be synthesized effectively. |
|
 121st General Meeting of the KCS
121st General Meeting of the KCS
 121st General Meeting of the KCS
121st General Meeting of the KCS