|
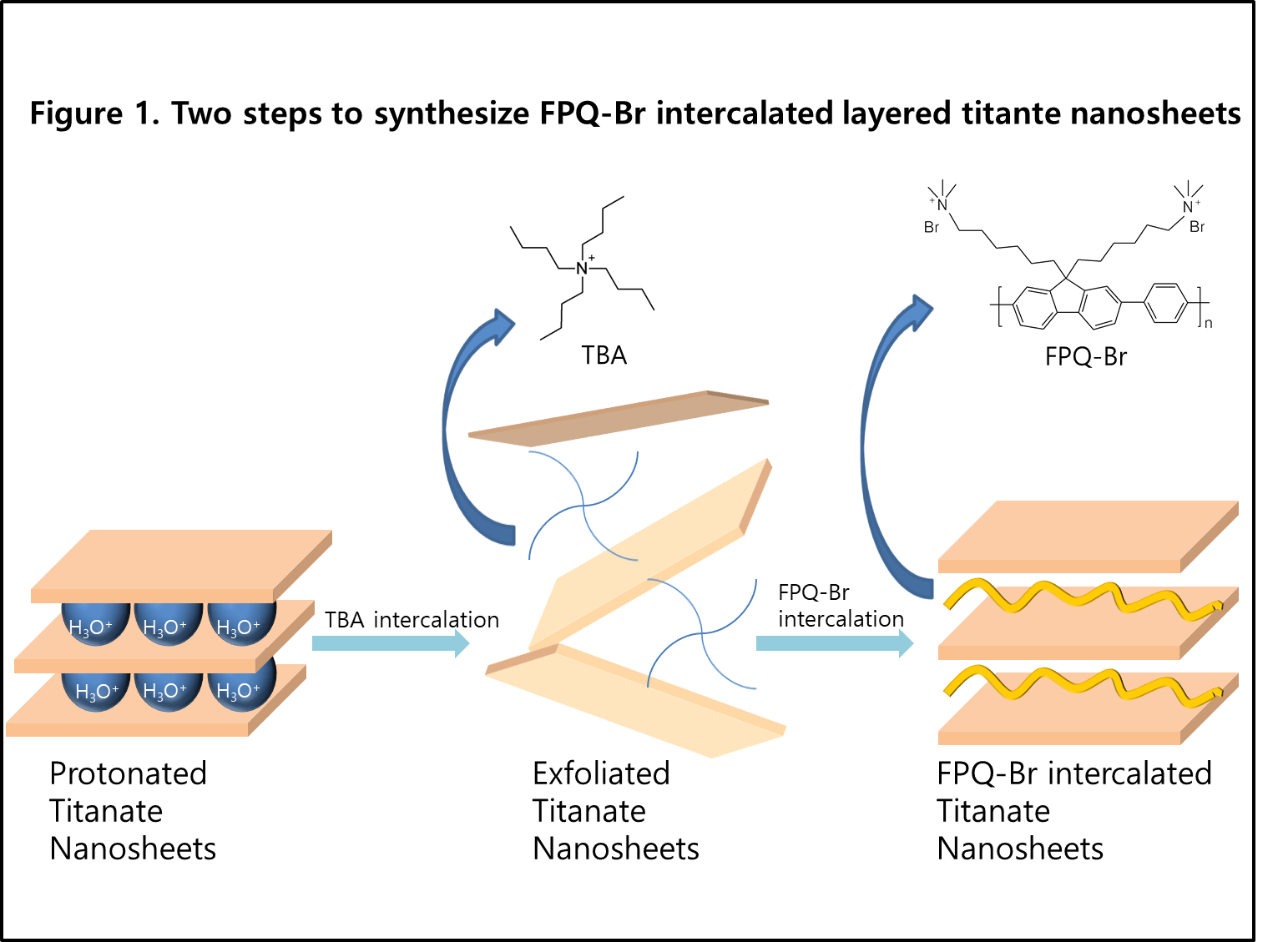
The intercalation of cationic compounds into layered titanium oxide semiconductors allows facile synthesis for the applications of Li batteries, biosensor, p-n heterojunction semiconductors and photocatalysts. However, titanium oxide only absorb in UV region of sunlight and this limitation decrease in the use of titanium oxides as photocaltalyst. Therefore, hybridizing of visible light absorbing materials with titanium oxides has gained great attention because these hybrid materials could be able to harvest the wider range of sunlight and show better photocaltalytic efficiency. Especially, intercalation of conducting polymer into the space between titanate nanosheets offers several advantages. Although cationic dyes can be easily intercalated through ion exchange reactions, they can be easily released at electrolyte solution. Furthermore, they are generally weak in photobleaching. In comparison, cationic polymers are more stable at sunlight and strongly binding with negatively charged titanium oxide layer due to their positively charged repeating units, so they cannot be easily released in electrolyte solution. Here, we report several steps to synthesis FPQ-Br intercalated layered titante nanosheets. The resulting hybrid materials have been being characterized by X-ray diffraction(XRD), Fourier transform infrared spectroscopy(FTIR), UV-vis spectroscopy, Thermogravimetry(TG), Cyclic voltametry(CV) and Scanning electron microscopy(SEM). Electrochemical impedance spectroscopy(EIS) and Metylene blue decomposition test in visible light will be measured to identify visible light photocatalytic properties. |
|
 121st General Meeting of the KCS
121st General Meeting of the KCS
 121st General Meeting of the KCS
121st General Meeting of the KCS