|
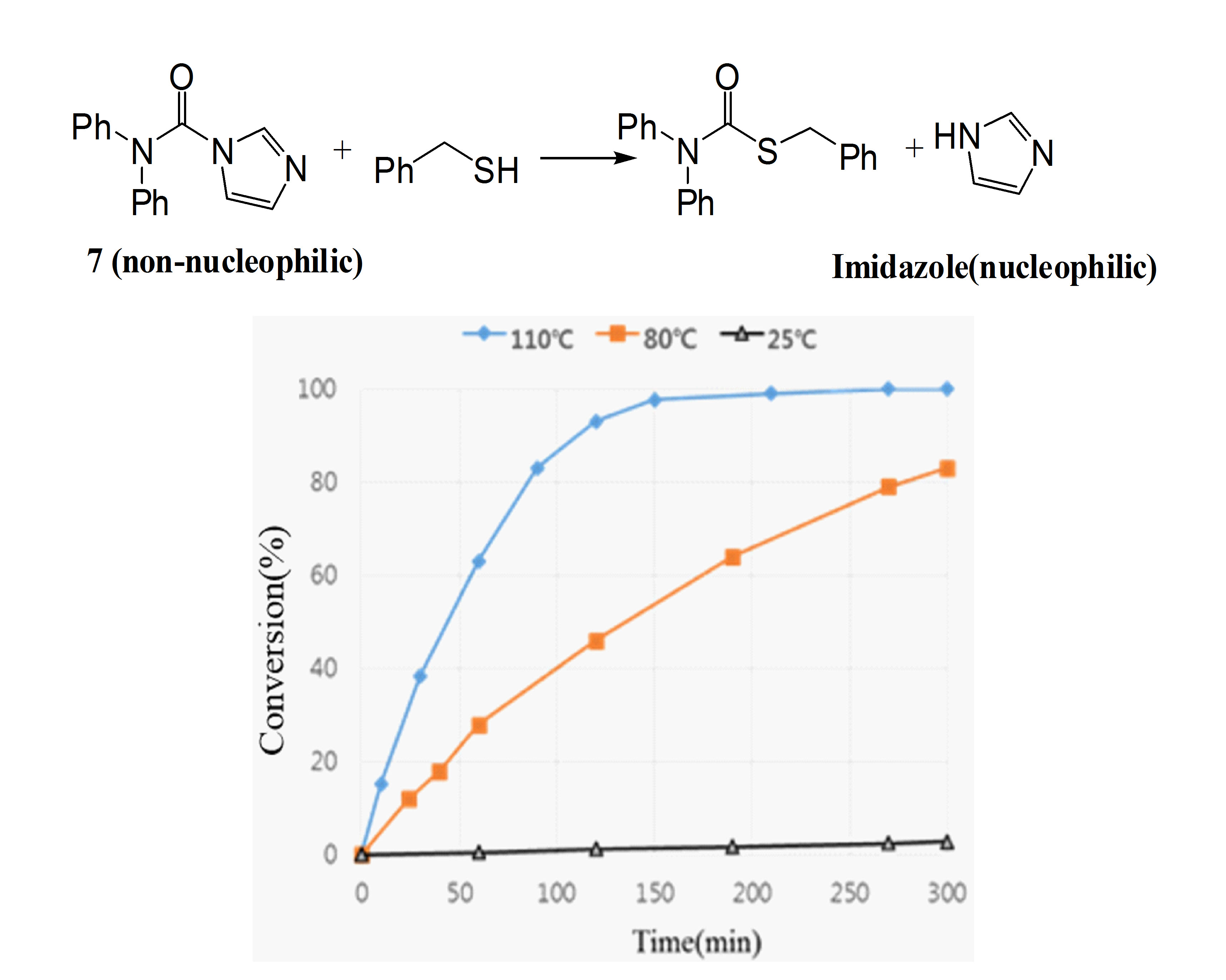
Sulfone containing polymer have high Abbe’s number and high refractive index. In the polymerization reaction of thiol and vinyl sulfone, imidazole act as a nucleophilic catalyst. However, unmodified imidazole can cure at room temperature, requiring lower temperature storage of resin formulations to prolong their pot-life. In this study, electron withdrawing group such as carbonyl group introduced to imidazole to decrease nucleophilicity of imidazole. Carbonylated imidazole is inert toward to vinyl sulfone group. Therefore, the viscocity of the monomer mixture can be controlled, giving enough time for homogenizing and degassing procedure. Imidazole derivatives should readily release active imidazole catalyst at a high temperature when attacted with a thiol monomer. In model study, modified imidazoles were evaluated by reaction with benzyl mercaptan and applied to polymerization of polythiol and divinyl sulfone. |
|
 122nd General Meeting of the KCS
122nd General Meeting of the KCS
 122nd General Meeting of the KCS
122nd General Meeting of the KCS