|

|
| Type |
Poster Presentation |
| Area |
Organic Chemistry |
| Room No. |
Grand Ballroom |
| Time |
10월 18일 (목요일) 11:00~12:30 |
| Code |
ORGN.P-70 |
| Subject |
Folding structure owing to introduction of an α-amino acid into the cis-2-aminocyclohexanecarboxylic acid oligopeptide |
| Authors |
Seung Chan Yoo, Younghun Kim1, moon-gun Choi1, Soo Hyuk Choi1,*
Yonsei University, Korea
1Department of Chemistry, Yonsei University, Korea
|
| Abstract |
|
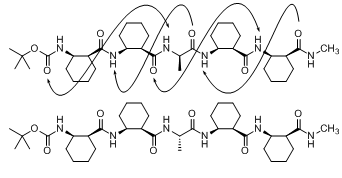
The cis-2-aminocyclohexanecarboxylic acid (cis-ACHC) oligopeptides with alternating chirality have been known to display both right- and left-handed 12/10-helical conformations in solution.
We synthesized several cis-ACHC pentapeptides of which the third residue is substituted with a simple α-amino acid such as alanine, valine, and 2-aminoisobutyric acid (Aib). Then, we analyzed how introduction of the α-amino acid into the cis-ACHC pentapeptide can alter its folding structure by using the CD, 2D-NMR and the single-crystal XRD methods.
Herein we demonstrate that introduction of D-alanine or D-valine can promote an intramolecular hydrogen bond to form a left-handed helical structure in this cis-ACHC pentapeptide. In contrast, introduction of L-alanine or Aib can interfere helical folding. |
|
|
| E-mail |
ysc7836@yonsei.ac.kr |
|
 122nd General Meeting of the KCS
122nd General Meeting of the KCS
 122nd General Meeting of the KCS
122nd General Meeting of the KCS