|
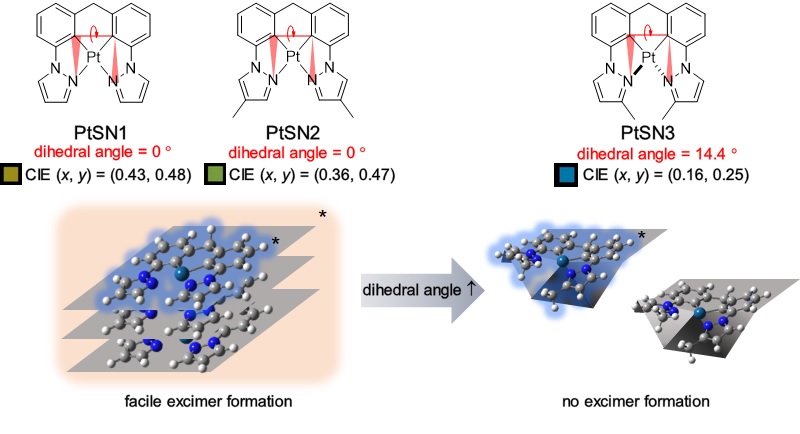
Square-planar d8 Pt(II) complexes constitute important dopants for organic light-emitting devices (OLEDs). However, blue electrophosphorescence from Pt(II) complexes remains underdeveloped. This lag originates from the deterioration of color purity due to the formation of excimers. One appealing approach to addressing this challenge would be breaking planarity of a Pt(II) complex using a tetradentate ligand having large steric hindrance. To achieve this goal, we designed and synthesized three Pt(II) complexes having tetradentate ligands with varied methyl substituents, di(3-(pyrazol-1-yl)phenyl)methide (for PtSN1), di(3-(4-methylpyrazol-1-yl)phenyl)methide (for PtSN2), and di(3-(3-methylpyrazol-1-yl)phenyl)methide (for PtSN3). The complexes displayed blue phosphorescence emission with peak wavelengths of 438-442 nm in dilute solutions. PtSN3 retained its inherent blue emission when doped in mCBP films at a concentration of 8 wt %, whereas PtSN1 and PtSN2 showed dominant excimer emissions at the same doping concentrations. The equilibrium constant (K) for the formation and dissociation of excimers were determined to be as large as 1.17 x 104 M-1 and 0.69 x 104 M-1 for PtSN1 and PtSN2, respectively. OLEDs were fabricated using the PtSN compounds as dopants, which yielded the maximum external quantum efficiencies of 14.1% (PtSN1), 13.7% (PtSN2), and 8.6% (PtSN3). The inferior performance of the PtSN2 device was ascribed to a low photoluminescence quantum yields (0.33 vs 0.57 (PtSN1) or 0.58 (PtSN2)), suggesting a trade-off relationship between the color purity and efficiencies. |
|
 123rd General Meeting of the KCS
123rd General Meeting of the KCS
 123rd General Meeting of the KCS
123rd General Meeting of the KCS