|
Introduction
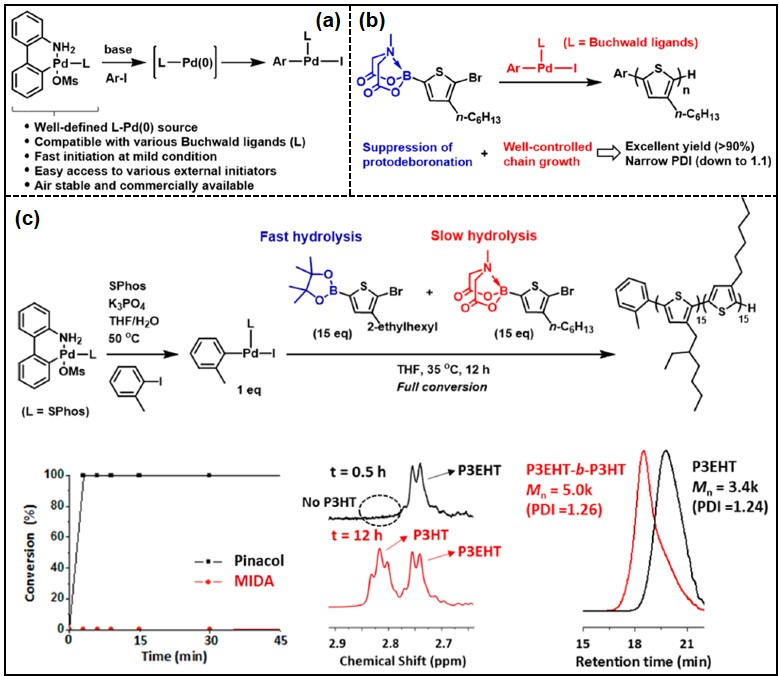
Kumada catalyst-transfer polycondensation (KCTP) method is the most widely used method to synthesize conjugated polymers, which have been intensively investigated due to their intriguing applications to optoelectronic devices, in a chain-growth manner. However, using moisture-sensitive Grignard reagents gave practical disadvantages. On the other hand, air-/moisture-stable boronates have been used for Suzuki-Miyaura catalyst-transfer polycondensation (SCTP), which makes SCTP as a promising alternative for KCTP. Despite these advantages, the use of a less efficient catalyst system and the competing protodeboronation of heterocyclic boronate monomer limited the wide application of SCTP.
Herein, we report a highly controlled SCTP of 3-alkylthiophenes using Buchwald dialkylbiarylphosphine Pd G3 precatalysts and N-methylimidodiacetic (MIDA)-boronates.
Results and Discussion
RuPhos and SPhos Pd G3 precatalysts were identified as excellent catalyst systems for the catalyst-transfer process by optimizing a small molecule model reaction. SCTP was tested using pinacol-boronate thiophene monomer, but controlled poly(3-hexylthiophene) (P3HT) was obtained for a low DP only. To improve the control, slowly hydrolyzed MIDA- boronate thiophene monomer was introduced. As a result, P3HT and P3EHT (up to 17.6 kg/mol) were prepared with excellent control, narrow dispersity (<1.32), and high yield (>90%).
Detailed mechanistic studies using NMR and MALDI-TOF spectroscopy revealed that both catalyst-transfer of Buchwald catalysts and slow hydrolysis of MIDA-boronate were crucial to achieve successful SCTP of P3HT. Furthermore, block copolymers were prepared via SCTP by both sequential monomer addition and one-shot methods.
|
|
 123rd General Meeting of the KCS
123rd General Meeting of the KCS
 123rd General Meeting of the KCS
123rd General Meeting of the KCS